This document is primarily intended for device labelers and provides information necessary for submitting data to the global unique device identification database gudid.
Fda unique device identification regulation.
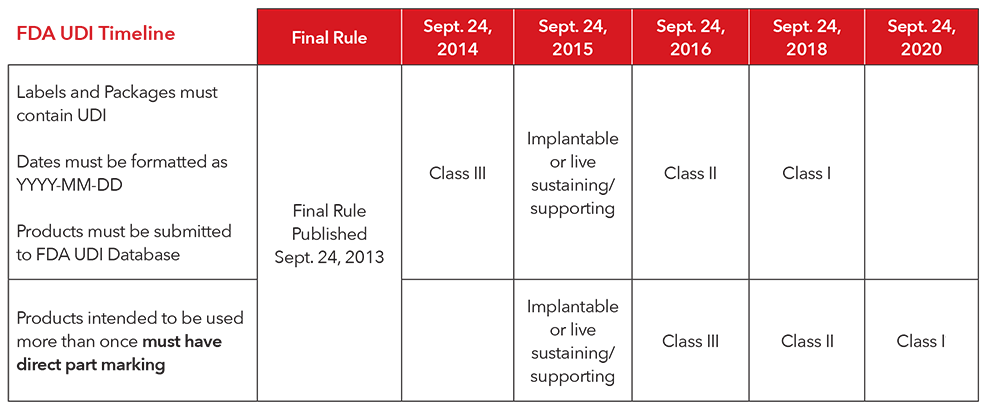
The us food and drug administration fda released in september 2013 a udi rule which establishes a udi system applying to all medical devices placed on the us market.
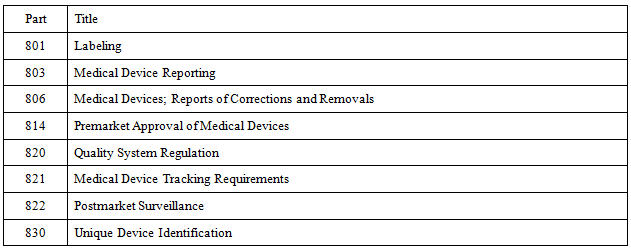
830 220 termination of fda service as an issuing agency.
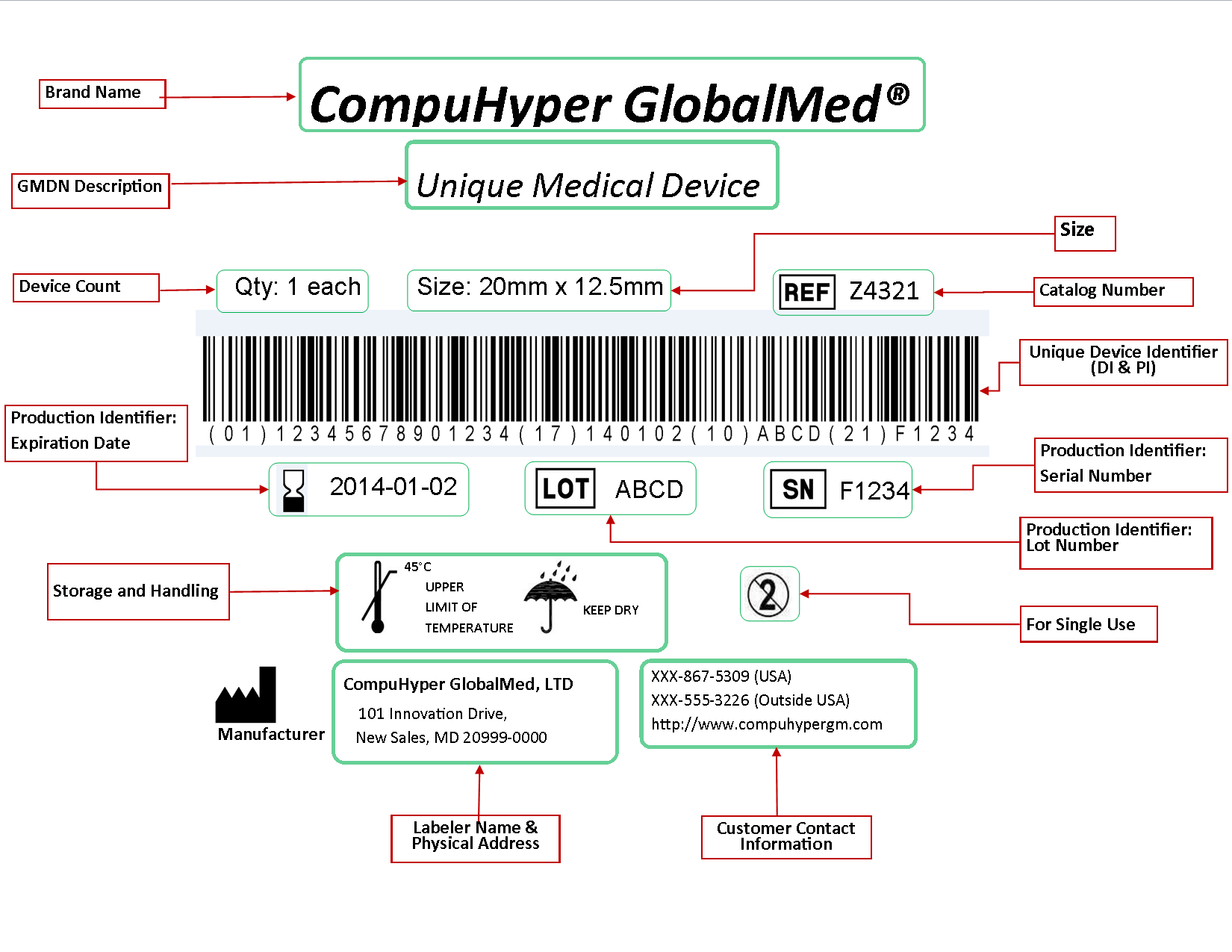
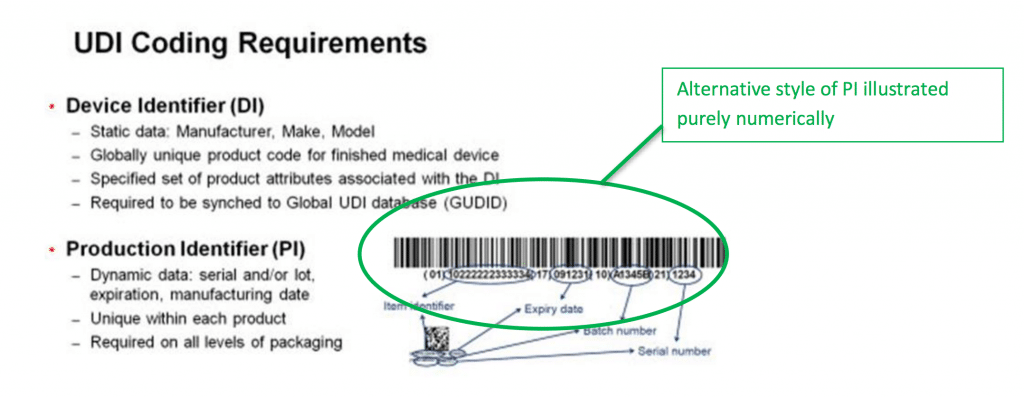
The unique device identification udi is a system used to mark and identify medical devices within the healthcare supply chain.
This document will assist industry particularly labelers as defined under 21 cfr 801 3 and fda staff in understanding fda s requirements for direct marking of devices for unique device.
830 310 information required for unique device identification.
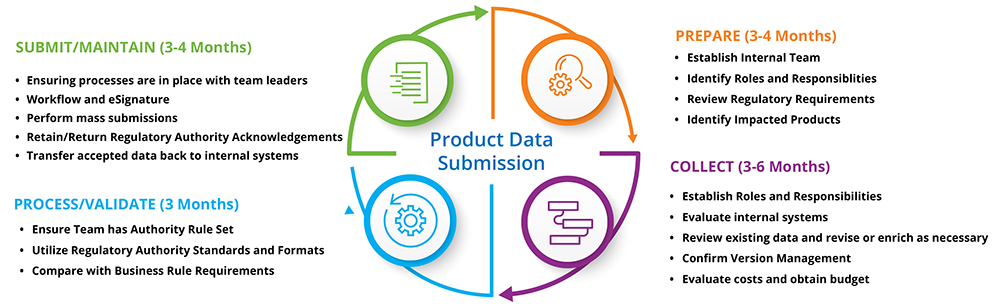
Unique device identification system.
Form and content of the unique device identifier udi draft guidance for industry and food and drug administration staff 08 14 2016 gudid submission.
Gs1 standards meet the us fda s criteria for issuing udis.
The fda established the unique device identification system to adequately identify medical devices sold in the united states from manufacturing through distribution to patient use.
The imdrf international medical device regulator forum the united states food and drug administration fda and the european commission are aiming for a globally harmonised and consistent approach to increase patient safety and help optimise patient care by.
Subpart e global unique device identification database 830 300 devices subject to device identification data submission requirements.
A draft version of this.
The global unique device identification database gudid is a database administered by the fda that will serve as a reference catalog for every device with a unique device identifier udi.
On 17 december 2013 gs1 has been accredited by the us fda as issuing agency for unique device identifiers udis.
































.jpg?width=1245&name=3%20COMMON%20MISCONCEPTIONS%20ABOUT%20MEDICAL%20DEVICE%20PACKAGING%20(1).jpg)



